1 phenylethanol density
Home » chemistry » 1 phenylethanol density1 phenylethanol density
1 Phenylethanol Density. ILO International Chemical Safety Cards ICSC 366. Substantial literature is found concerning the use of glutaraldehyde for protein immobilization yet there is no agreement about the main reactive. The electron density on the carbonyl carbon atom decreases and this favours the nucleophile attack. 760 mm Hg at 2772 F NTP 1992 National Toxicology Program Institute of Environmental Health Sciences National Institutes of Health NTP.
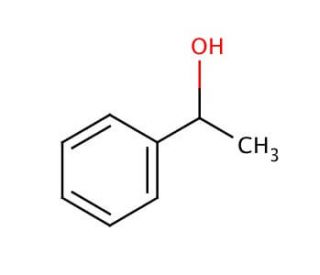 1 Phenylethanol Cas 98 85 1 Scbt Santa Cruz Biotechnology From scbt.com
1 Phenylethanol Cas 98 85 1 Scbt Santa Cruz Biotechnology From scbt.com
Predict the products of the following reactions. Substantial literature is found concerning the use of glutaraldehyde for protein immobilization yet there is no agreement about the main reactive. The existence of this group in the ortho position decreases the electron density in the O H bond. Thermophysical Properties Gosudarstvennoe Energeticheskoe Izdatelstvo Moscow 1963. Isoamyl alcohol is a colorless liquid with the formula C 5 H 12 O specifically H 3 C 2 CHCH 2 CH 2 OH. Iii Pentan-l-ol using a suitable alkyl halide.
Substantial literature is found concerning the use of glutaraldehyde for protein immobilization yet there is no agreement about the main reactive.
Site directed mutants were constructed in cytochrome p450cam to re-engineer the stereochemistry and coupling of ethylbenzene hydroxylation. Thermophysical Properties Gosudarstvennoe Energeticheskoe Izdatelstvo Moscow 1963. It occurs widely in nature being found in a variety of essential oilsIt has a pleasant floral odor. In contrast a methoxy group is an electron releasing group. Consequently it is easier to give away a proton. As one of these fragmentsalong with the molecule as a wholeexhibits accordance.
 Source: scbt.com
Source: scbt.com
ILO International Chemical Safety Cards ICSC 366. Consequently it is easier to give away a proton. In contrast a methoxy group is an electron releasing group. It is also known as isopentyl alcohol isopentanol or in the IUPAC recommended nomenclature 3-methyl-butan-1-olAn obsolete name for it was isobutyl carbinol. Give two reactions that show the acidic nature of phenol.

7487discordant may indicate variable stability. Density of liquid D2O in gcm3 at a pressure of 100 kPa 1 bar. Differences in the status of disordered fragments 2445accordant. Furthermore the o -nitrophenoxide ion formed after the loss of protons is stabilized by resonance. 760 mm Hg at 2772 F NTP 1992 National Toxicology Program Institute of Environmental Health Sciences National Institutes of Health NTP.
Furthermore the o -nitrophenoxide ion formed after the loss of protons is stabilized by resonance. For instance 987 selectivity to 1-phenylethanol in the hydrogenation of acetophenone was achieved over 02Ptm-Al 2 O 3-H 2 which is higher than 02Ptp-Al. The existence of this group in the ortho position decreases the electron density in the O H bond. Furthermore the o -nitrophenoxide ion formed after the loss of protons is stabilized by resonance. It is one of several isomers of amyl alcohol pentanol.
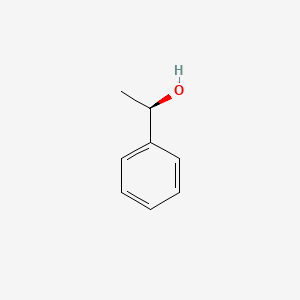
 Source: molinstincts.com
Source: molinstincts.com
It can be present in at least 13 different forms depending on solution conditions such as pH concentration temperature etc. 7487discordant may indicate variable stability. Consequently it is easier to give away a proton. The reactions showing acidic nature of phenol are. Furthermore the o -nitrophenoxide ion formed after the loss of protons is stabilized by resonance.
 Source: sigmaaldrich.com
Source: sigmaaldrich.com
Compare its acidity with that of ethanol. As one of these fragmentsalong with the molecule as a wholeexhibits accordance. It can be present in at least 13 different forms depending on solution conditions such as pH concentration temperature etc. It is one of several isomers of amyl alcohol pentanol. 7487discordant may indicate variable stability.
 Source: en.wikipedia.org
Source: en.wikipedia.org
Glutaraldehyde possesses unique characteristics that render it one of the most effective protein crosslinking reagents. Occupational Safety and Health Administration OSHA 3210 Vapor Pressure. 760 mm Hg at 2772 F NTP 1992 National Toxicology Program Institute of Environmental Health Sciences National Institutes of Health NTP. Substantial literature is found concerning the use of glutaraldehyde for protein immobilization yet there is no agreement about the main reactive. The reaction with the wild type enzyme produces one regioisomer 1-phenylethanol with 5 reduced nicotinamide adenine deoxyribonucleic acid product conversion of and a ration of 7327 for the R and S enantiomers respectively.

Ii cyclohexylmethanol using an alkyl halide by an SN 2 reaction. Site directed mutants were constructed in cytochrome p450cam to re-engineer the stereochemistry and coupling of ethylbenzene hydroxylation. Compare its acidity with that of ethanol. It is one of several isomers of amyl alcohol pentanol. The electron density on the carbonyl carbon atom decreases and this favours the nucleophile attack.
 Source: chemsynthesis.com
Source: chemsynthesis.com
Thermophysical Properties Gosudarstvennoe Energeticheskoe Izdatelstvo Moscow 1963. I 1 -phenylethanol from a suitable alkene. In contrast a methoxy group is an electron releasing group. 7487discordant may indicate variable stability. 5 mm Hg at 570 F.

Glutaraldehyde possesses unique characteristics that render it one of the most effective protein crosslinking reagents. The reactions showing acidic nature of phenol are. 7487discordant may indicate variable stability. Compare its acidity with that of ethanol. It is also known as isopentyl alcohol isopentanol or in the IUPAC recommended nomenclature 3-methyl-butan-1-olAn obsolete name for it was isobutyl carbinol.
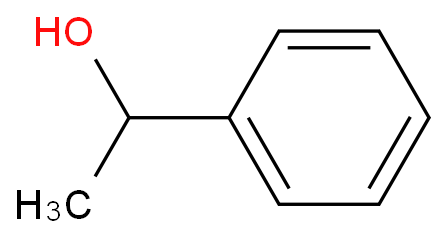 Source: guidechem.com
Source: guidechem.com
The electron density on the carbonyl carbon atom decreases and this favours the nucleophile attack. It is one of several isomers of amyl alcohol pentanol. Kirillin VA Ed Heavy Water. Hence it increases the. Relative vapor density air 1.
If you find this site serviceableness, please support us by sharing this posts to your own social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title 1 phenylethanol density by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.